 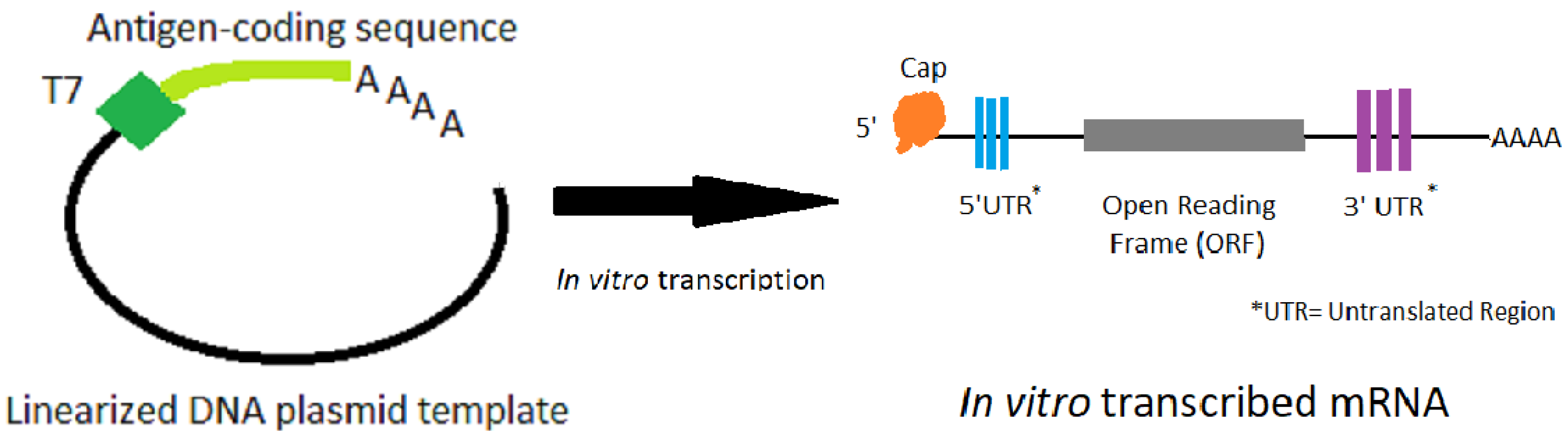
However, it is important to emphasize that we are still early in the Over the last four years, we have started 9 clinical trials for mRNA This includes successful early-stage (Phase 1) clinical trials against five other respiratory viruses (two pandemic influenza strains, RSV, hMPV, and PIV3). Over the past few years, we have demonstrated its potential in vaccines across more than 1,000 subjects in ourĬlinical trials. How well validated is your mRNA platform? The open-label trial is expected to enroll 45 healthy adult volunteers ages 18 to 55 years over approximately six weeks.įrequently asked questions about our technology & platform Immunogenicity means the ability of the vaccine to induce an immune response in participants. This Phase 1 study will provide important data on the safetyĪnd immunogenicity of mRNA-1273. On March 16, 2020, the NIH announced that the first participant in its Phase 1 study for mRNA-1273 wasĭosed, a total of 63 days from sequence selection to first human dosing. New Drug (IND) application filed by the NIH for mRNA-1273 and allowed the study to proceed to begin clinical trials. Food and Drug Administration (FDA) completed its review of the Investigational On February 24, 2020, the clinical batch was shipped from Moderna to the NIH for use in their Phase 1  
The batch then proceeded to analytical testing for release. Of 25 days from sequence selection to vaccine manufacture. On February 7, 2020, the first clinical batch, including fill and finishing of vials, was completed, a total Manufacture of this batch was funded by the Coalition for Epidemic Preparedness Innovations (CEPI). At that time, the National Institute of Allergy and Infectious Diseases (NIAID), part of NIH, disclosed their intent to run a Phaseġ study using the mRNA-1273 vaccine in response to the coronavirus threat and Moderna mobilized toward clinical manufacture. Research team finalized the sequence for mRNA-1273, the Companys vaccine against the novel coronavirus. National Institutes of Health (NIH) and Modernas infectious disease On January 11, 2020, the Chinese authorities shared the genetic sequence of the novel coronavirus. Our work to advance our vaccine candidate (mRNA-1273) and responds to frequently asked questions. Moderna is proud to be among the many groups working to respond to this continuing global health emergency. Relying on vaccines based on the original Covid strain for autumn booster programmes will result in “a shorter duration of protection, less robust protection and more severe disease and hospitalisation”, Burton said.Modernas Work on a Potential Vaccine Against COVID-19 The new vaccine combines 25 micrograms of the original Moderna Covid vaccine with 25 micrograms of vaccine specifically targeted at the Omicron variant.ĭr Paul Burton, Moderna’s chief medical officer, said the company is ready to deliver the vaccine for booster programmes as early as August. The company released data to show the vaccine, called mRNA1273.214, protects against the Omicron strains BA.4 and BA.5, which are driving a current surge of cases. Separately, Moderna said it plans to apply within days to US and UK regulators for approval for its new two-strain Covid-19 vaccine. Moderna said it planned to expand its presence in the UK through investments in R&D and promised deliver a large share of its clinical trials in the UK. The health secretary, Sajid Javid, added: “Our new partnership with Moderna will cement the UK’s status as a science superpower, significantly boosting the economy and creating jobs – and it has the potential to unlock the next generation of cutting-edge vaccines to fight diseases such as Covid, seasonal flu and RSV.” He added: “We’ve all seen what vaccines can do, and today’s partnership brings us one step closer to finding cures for some of the most devastating diseases.” Boris Johnson said the investment would “guarantee jabs in arms against some of the toughest viruses out there, bringing us to the forefront of the fight against future threats”. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
